 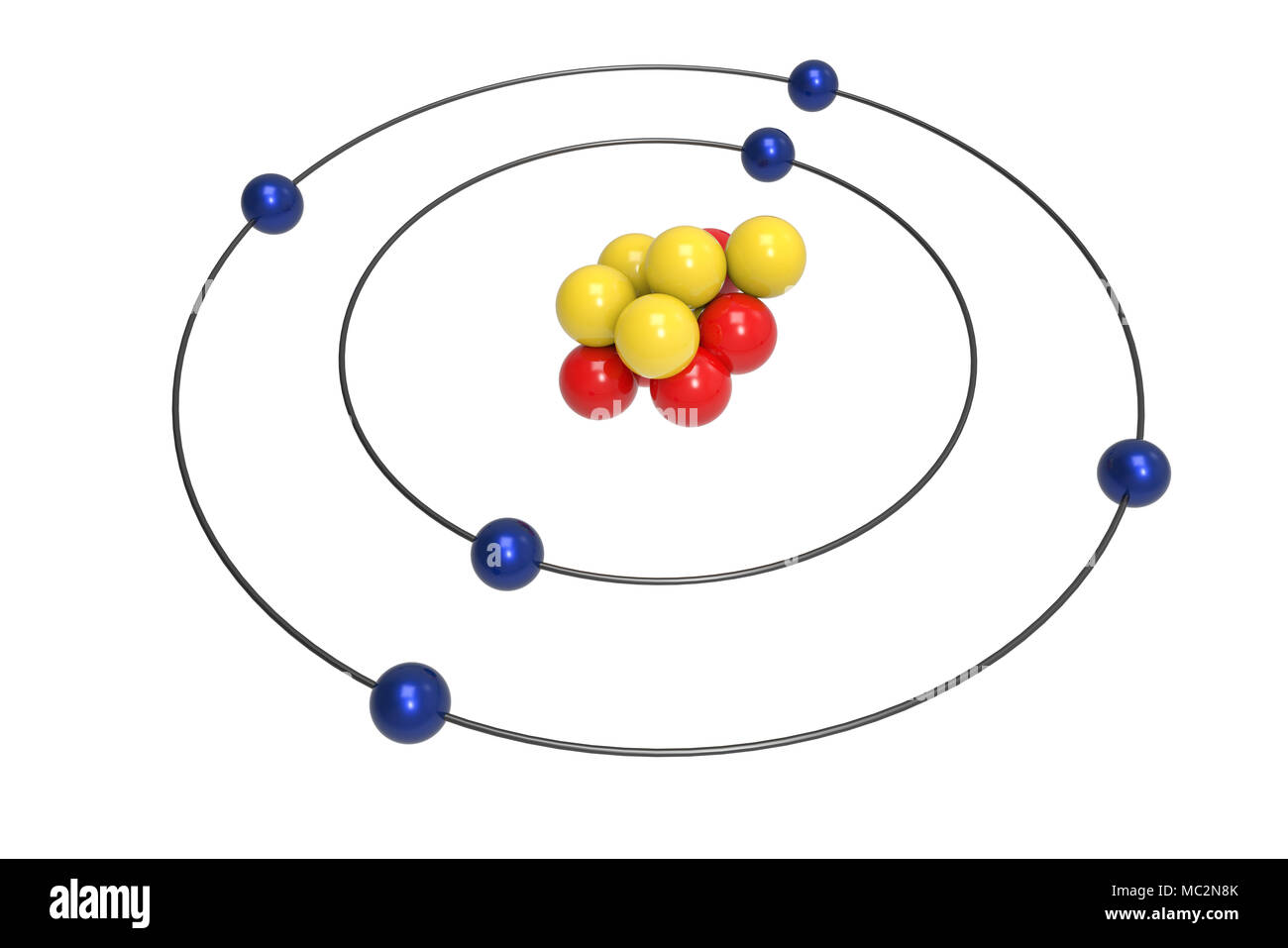 
You should have 6 total electrons for Carbon. Again, write e- and the number of electrons. C P+ = 6 N0 = 6 These go in the 2nd shell. How many electrons are in the outer shell? C P+ = 6 N0 = 6 2 e-Ĩ Bohr Diagrams Since you have 2 electrons in the first shell, you need to add 4 more. Write the number of electrons that shell holds and e- for electrons an their charge. The first shell can only hold 2 electrons. C P+ = 6 N0 = 6ħ Bohr Diagrams C Add the electrons. Shells are named with letters: 1 = K shell = 2 electrons 2 = L shell = 8 electrons 3 = M shell = 8 electrons 4 = N shell = 18 electrons 5-7 = O, P, Q = up to 32 electrons Remember: The outer shell of an atom (no matter what letter) can only hold 8 electrons!Ħ Bohr Diagrams Carbon is in the 2nd period, so it has two energy levels, or shells. C P+ = 6 N0 = 6 Note: Round mass to nearest 1 when figuring neutrons.ĥ The electron shells surrounding the nucleus each hold a particular number of electrons. Put a + by the P and a little 0 by the N. Write the number of Protons and Neutrons the element has inside the nucleus. Elements in the 2nd period have two energy levels, and so on.Ĥ Bohr Diagrams C Draw a nucleus with the element symbol inside. This is how many electrons you will draw.ģ Bohr Diagrams Find out which period (row) your element is in.ġ 2 3 4 5 6 7 Elements in the 1st period have one energy level. 2 Bohr Diagrams Find the element on the periodic table.ĭetermine the number of electrons-it is the same as the atomic number. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
